Modern Canna’s Laboratory Director, Jini Curry, recently published an article on Analytical Cannabis – a leading cannabis science publication for scientists, technicians, and business professionals working in the cannabis, hemp, and psychedelics industries. This article provides an overview of Jini’s article, along with the full article linked towards the bottom.
Both yeast and mold are spore-producing microorganisms that feed on organic matter, thus classifying them as fungi. Various species of yeasts and molds have a strong affinity to grow on plant material, specifically in humid, warm conditions. Hence, it is widely necessary and applicable to be testing cannabis plants for yeast and mold in order to ensure patients and users do not consume these harmful microbes.
To test cannabis plants for this, total yeast and mold (TYM) counts are performed. TYM tests are used to detect and quantify the amount of fungal growth on the given plant material. There are currently two different processes used to do so: quantitative polymerase chain reaction (qPCR) and/or traditional plating methods.
A dual of methodologies: qPCR vs. Traditional Plating
qPCR has been found to be an effective option in determining certain fungal species, since the DNA of the sample input is quantified. It is also useful in certain scenarios because certain fungal species exhibit resistance to growing on plates. However, false negatives and low quantitation/under estimation have been found with qPCR. When this happens, traditional plating methods can be turned to and utilized.
Thus, a grey area has been formed and consistency seems to lack with TYM testing. Cannabis regulators and laboratory personnel are stuck in the middle. Regardless of the method, it comes down to accuracy. Laboratories must produce accurate, verifiable, and reproducible data – for all tests and matrices, not just TYM.
So which method is the most accurate?
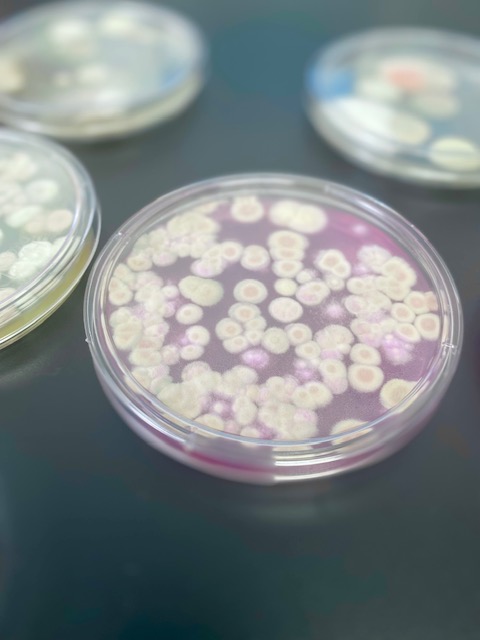
In an effort to get to the bottom of this burning question, Modern Canna took a TYM investigation into their own hands. Our scientists ran into a discrepancy from that start. Upon examining two different plating materials and their TYM reference values, a major variation was found. The certified activity is 152,000 colony forming units per gram (CFU/g) when Petrifilm Rapid Yeast/Mold Plates (P-RYM) are used, and 423,000 CFU/g when Sabouraud Dextrose (SabDex) Agar is the growth medium. This extreme difference raised a red flag, along with a lot of unanswered questions.
Now a mystery, Modern Canna decided that the next step surrounding TYM was to test cannabis samples on multiple different growth mediums AND via qPCR to see how the results lined up with one another.
3, 2, 1….Cross examination
An initial study was conducted by analyzing ten cannabis flower samples for TYM using plating methods on various growth mediums (listed below) and qPCR technology.
- Compact Dry (CD)
- Petrifilm Rapid Yeast and Mold (P-RYM)
- Petrifilm Non-Rapid Yeast and Mold (P-NRYM)
- Potato Dextrose Agar (PDA)
- Potato Dextrose Agar with Chlortetracycline (PDAC)
- Dichloran Rose Bengal Chlorampenicol (DRBC)
- Sabouraud Dextrose (SabDex)
However, the results obtained from this experiment were unfortunately inconclusive. This was due to the large variations across the methods and the lack of reproducibility in results.
Back to the drawing board
In order to implement more control, our laboratory decided to start over and conduct two separate experiments.
- Experiment #1 – Used a single sample and analyzed it five times at three different dilutions.
- Experiment #2 – Looked at six different samples, processed in triplicate, (three of which were treated with radiation) at the same dilutions.
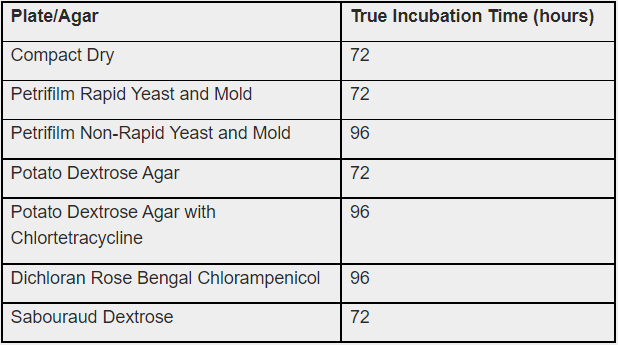
In the table below are the conclusions obtained from the experiment, which suggest incubation times for the various growth mediums used:
Jini stated, “the study revealed that while TYM numbers sometimes align from method to method, it is more common that the results will vary drastically, sometimes by more than 50%. This is one of the main pitfalls that cannabis methodologies face. Cannabis is a complex organic material that changes from sample to sample”. From vapes, to edibles, to flower, to tinctures – the variety of cannabis matrices added a deeper layer of complexity to this standardization issue.
What exactly does this mean for the future of cannabis TYM testing?
Our findings revealed that the typical dilutions performed in plating methods may cause data to be skewed. Whereas, the qPCR may lead to false-negative results if there is too much contamination or if certain microbes are present.
Below are the additional conclusions:
- Typical dilutions performed in plating methods may cause data to be skewed
- Incubation time plays a pivotal role in the results obtained
- Most agars used did not show complete colony growth in cannabis until at least 72 hours, even though some of the recommended incubation times are 48 hours
- The qPCR may lead to false-negative results if there is too much contamination or if certain microbes are present.
All in all, the lack of consistency suggests that more concise data validation actions need to be implemented. Jini concluded that “it is our lab’s suggestion that the industry consider moving away from including TYM as part of the standard cannabis testing panels and instead, test for additional yeast or mold species that commonly grow on cannabis and are dangerous to humans”. Some of these microbes include:
- Aspergillus species
- Botrytis (bud rot)
- Powdery mildew
- Fusarium species (root rot)
- Other Penicillium species
In Conclusion…
There are clearly pros and cons to both plating and qPCR methodology. While TYM analysis may be effective in determining the cleanliness of a product’s processes, it may not be the best analysis to determine if products are safe for human consumption. Jini concluded that “the food and pharmaceutical industries have extensively researched how analyses such as TYM work for their products and ultimately, the cannabis industry needs to apply that same level of scrutiny to TYM in cannabis products”.
Jini’s Analytical Cannabis article, “The Benefits and Pitfalls of Total Yeast and Mold Counts in Cannabis Labs”, can be found here: Total Yeast and Mold in Cannabis: Benefits & Pitfalls – Analytical Cannabis



 Modern Canna
Modern Canna